Drugging inflammaging at the source
Drive inflammation, or fuel repair — the upstream switches.
Drive inflammation, or fuel repair — the upstream switches.
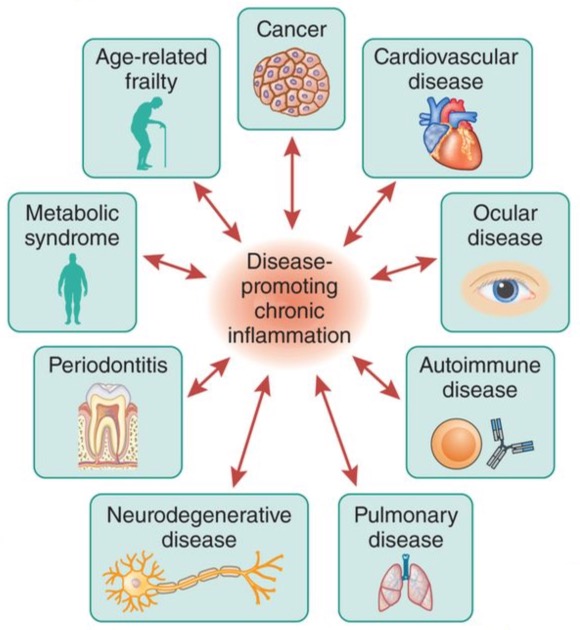
Inflammaging — chronic, low-grade inflammation that builds up as we age — is the shared upstream cause of Alzheimer's, heart disease, frailty, and most of the conditions that erode our quality of life as we age. Three failures in our cells generate it. Each one is a drug target.
The same broken support cells underlie diseases of aging across the body.
End-organ disease follows — neurodegeneration, sarcopenia, metabolic syndrome, heart dysfunction, atherosclerosis, arthritis, liver fibrosis, frailty. Adapted from Furman et al., Nat Med 2019.
7.2 million Americans live with Alzheimer's today. By 2050, it'll be nearly 13 million. Today's drugs are first steps — they are not enough.
There are 13 million Americans caring for someone with Alzheimer's right now — most of them quietly, often a daughter, often for years. We're building this for them.
Projected to nearly double — to 13 million — by 2050.
Often unpaid, often a daughter, often providing 24/7 care for years.
Projected to reach nearly $1 trillion per year by 2050.
More than breast cancer and prostate cancer combined.
Sources: Leqembi & Kisunla FDA labels; Alzheimer's Association 2025 Facts & Figures; AARP, 2024–25.
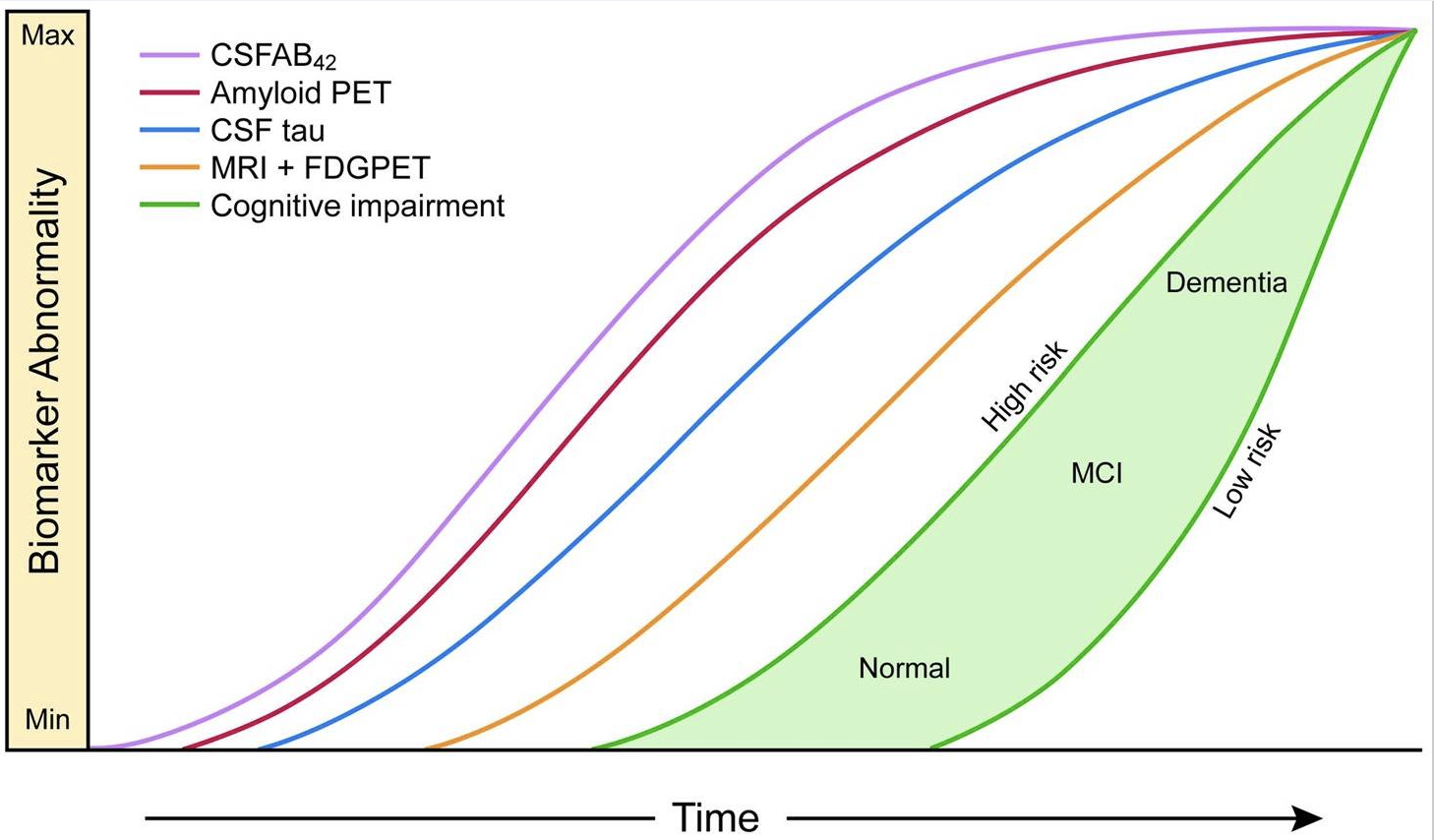
FDG-PET imaging captures glucose use across the brain. In people who later develop Alzheimer's, glucose use drops measurably — sometimes 10 years before any clinical symptoms appear.
It's a different signal from amyloid or tau scans. Those measure protein buildup. FDG-PET measures whether the brain is actually functioning — whether cells are getting fuel. That fuel failure is what we target.
Willow's lead candidate restores the brain's fuel-delivery system at the source — the astrocyte cell that feeds neurons. The same functional-biology + mouse-blockade development path that delivered Keytruda, Skyrizi, and Humira to humans.
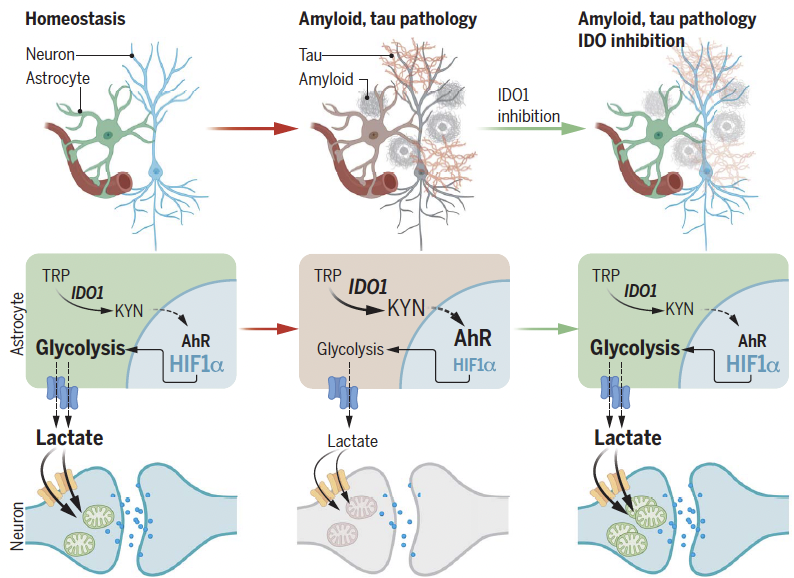
When it comes to brain function, neurons get a lot of attention. But healthy brains depend on the cooperation of many kinds of cells. The most abundant non-neuronal cells in the brain are astrocytes — star-shaped cells with a lot of responsibilities. They shape neural circuits, participate in information processing, and provide nutrient and metabolic support to neurons.
When inflammaging hits the brain, astrocytes lose their ability to fuel neurons. Amyloid plaques, tau tangles, and inflammatory cytokines flip on an enzyme in astrocytes called IDO1. Their fuel-making machinery collapses. Neurons starve. Cognition fades. Willow's lead drug turns IDO1 back off — and the fuel comes back online.
IDO1 is the first. Two more switches reach beyond the brain — into the inflammation amplifier in your blood, and the cleanup crew across every organ. One companion diagnostic visualizes them all.
Drug target: IDO1
Drug target: TREM1
Drug target: Program 3
Companion diagnostic: Innate-PET™
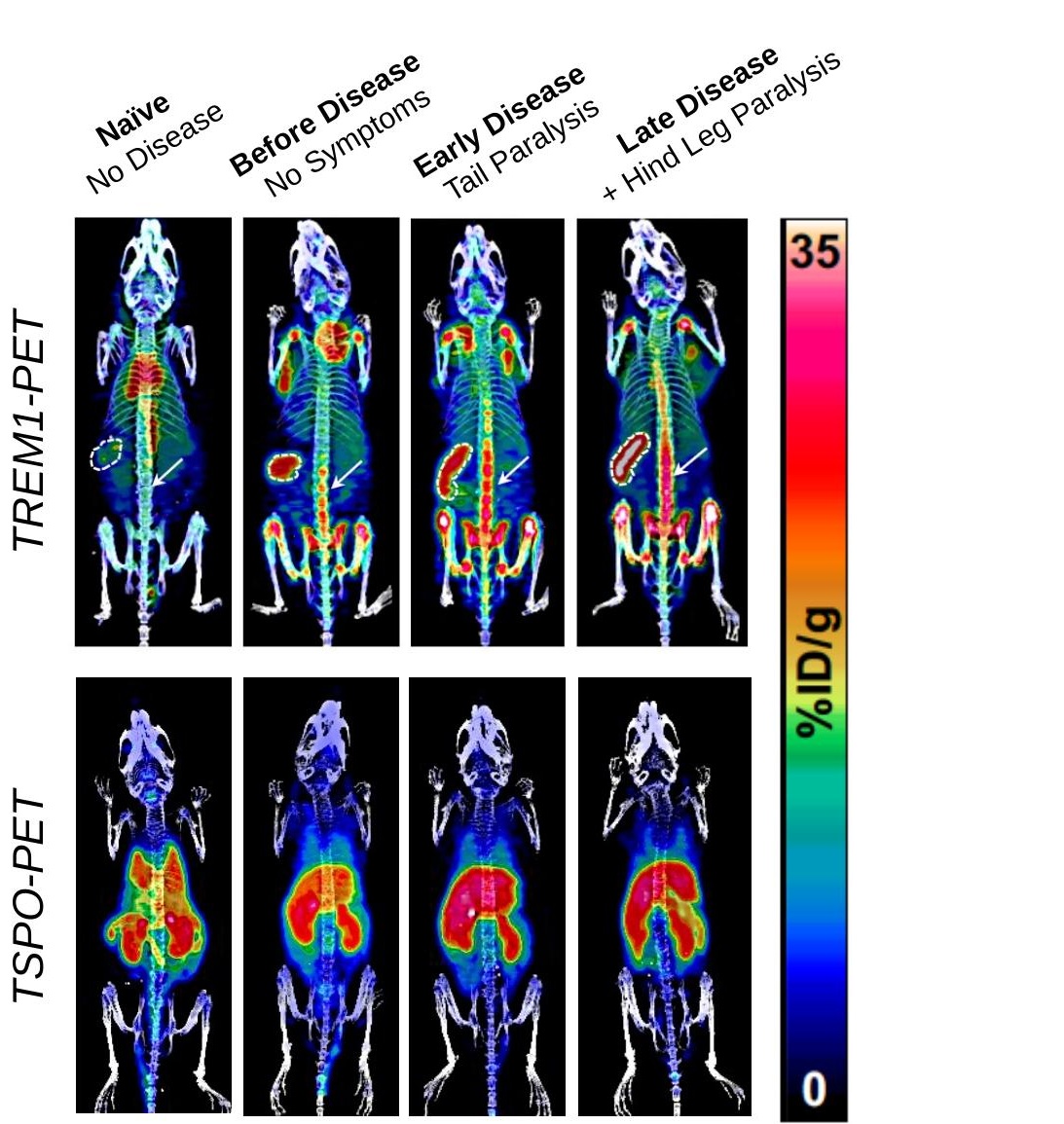
14–17× higher sensitivity than TSPO-PET in EAE — the current gold-standard inflammation tracer.
Tracks peripheral and CNS inflammation in one scan. Stratifies patients across IDO1, TREM1, and Program 3 trials with one proprietary diagnostic, reusable across the entire pipeline.
For each target, in each cell type, we have two independent kinds of evidence. Human genetics points at the cell. Mouse experiments confirm that fixing the target rescues disease.
When human genetics and mouse experiments agree on which cells to fix, the drug usually works. That's the same combination behind Keytruda (PD-1 in cancer), Skyrizi (IL-23 in psoriasis), Humira (TNF in rheumatoid arthritis), and the GLP-1 class.
Human genetics points at the cell types. Functional biology pinpoints the targets. The same kind of mechanism that PD-L1 followed in cancer.
Blocking IDO1 rescues cognition across multiple Alzheimer's mouse models. Replicated in human stem-cell astrocytes.
Minhas et al. — Andreasson lab. Covered by Nature, NPR, NIH.Switching off TREM1 in peripheral immune cells rescues cognition in aging and Alzheimer's mouse models. The drug never has to cross the blood-brain barrier.
Wilson, James, Andreasson et al. — Andreasson and James labs.Novel TREM1-PET tracer maps activated inflammation across the body. 14–17× higher signal than the current standard. Stanford has licensed this technology to Willow.
Chaney, Andreasson, James — James lab.Additional work connecting IDO1, TREM1, Innate-PET, and Program 3 to pan-organ aging is being prepared for publication.
Targeting glial metabolism early in the Alzheimer's cascade could be a promising therapeutic approach to prevent or delay disease progression.
An honest timeline of what's ahead. Each milestone is years of work.
IDO1 lead drug optimization and the safety studies the FDA requires before human trials. TREM1 and the cleanup-crew programs in optimization.
Phase 1a in healthy volunteers — confirms the drug is safe and reaches the brain at the dose we expect.
Phase 1b in people with mild cognitive impairment — measures whether the drug restores brain glucose use, the fuel signal we're targeting.
Phase 2 cognition and biomarker results. If positive, the path to FDA review opens.
A full technical pipeline is available on request for partners and investors.
We'll send a short, periodic update — major milestones, peer-reviewed publications, and clinical trial news. No spam. For families and caregivers, we also recommend the Alzheimer's Association for resources, support, and clinical trial matching via their TrialMatch service.
Plain answers to the questions we get most often.
Inflammaging is the chronic, low-grade inflammation that builds up in our bodies as we age — even when there's no infection or injury. It's not the kind of inflammation you feel when you sprain an ankle. It's silent, slow, and cumulative.
Researchers now recognize it as a major upstream contributor to many age-related diseases — Alzheimer's, heart disease, type 2 diabetes, frailty. Three failures in the body's first-responder immune cells and brain support cells generate it. Each one is a drug target.
Leqembi (Eisai) and Kisunla (Lilly) are antibody infusions that clear amyloid plaques. They were a milestone, but their practical limitations are real — see the Alzheimer's section above for the breakdown.
Willow's lead drug targets a different problem: the cells that fuel neurons stop working. We're building an oral pill that switches the brain's fuel system back on — complementary to amyloid drugs, not competitive.
Drug development is slow by design — the regulatory bar is high for good reason. Our current plan: first human safety trial in 2027, first Alzheimer's patient trial in 2028, efficacy readouts in 2030, and FDA review in the years after. If our results hold up through clinical trials, Willow's lead therapy could reach patients in the early 2030s.
The foundation: the science is already published in Science (2024) and Nature Neuroscience (2024), and the lab work has been replicated across multiple Alzheimer's mouse models and human stem-cell-derived brain support cells.
Not yet — we're not in human trials. When we open enrollment (planned 2027 for healthy volunteers, 2028 for early-stage Alzheimer's patients), we'll announce trial sites and eligibility criteria here, and you can sign up above to be notified.
In the meantime, the Alzheimer's Association TrialMatch service can connect families with currently-enrolling trials. ClinicalTrials.gov is the official US registry.
Alzheimer's is our lead indication because the science is most advanced there. But the same biology — failing brain support cells, an over-active inflammation amplifier in the bloodstream, and broken cleanup crews across organs — drives many age-related diseases.
We expect Willow's programs to expand into frontotemporal dementia, Parkinson's, and other inflammatory diseases of aging — once we've proven the approach in Alzheimer's first.
Three ways. First, subscribe to milestone updates — we'll let you know when trials open. Second, support the Alzheimer's research ecosystem broadly: the Alzheimer's Association and ADDF fund critical research, including some of ours. Third, if you're a researcher, clinician, partner, or potential collaborator, reach out directly.
Programs include Opdivo (nivolumab) · Erbitux · Erivedge · five clinical-stage PET tracers · IDO1 (Science 2024) and TREM1 (Nat Neurosci 2024) senior authorship.
Professor of Neurology, Stanford. Neuroinflammation KOL; NIH-funded 25+ years. Senior author IDO1 (Science 2024) and TREM1 (Nat Neurosci 2024).
Associate Professor of Radiology & Neurology, Stanford. Five PET tracers in clinic. Senior author Innate-PET (STM 2023). Roger Tsien Award 2024.
Chief, Memory Disorders Division, Stanford. 50+ AD / FTD trials as PI or site lead, including anti-amyloid pivotal studies.
Professor of Urology & Developmental Biology, Stanford. NAS member. Co-founder Fate Therapeutics, PellePharm. Co-inventor of Erivedge.
For partnerships, clinical collaborations, press, or to follow Willow's progress — get in touch.
We'll reply within 24 hours. Mention your area of interest and we'll set up a call.